Apple cider vinegar (ACV) is a popular ingredient in various food items like a salad dressing, dressings, pickles, and marinades. It is made by fermenting crushed apples, yeast, and sugar, which results in a liquid with numerous health benefits, such as supporting digestion, controlling high blood pressure and sugar levels, and aiding in weight loss. One of the properties that contribute to its health advantages is its pH level, which determines if the vinegar is more acidic or alkaline.
The pH level of apple cider vinegar falls between 2 and 3, making it an acidic substance. Some even make a claim that ACV can help to alkalize the body, but it’s essential to understand that the entire body maintains a slightly alkaline pH in the range of 7.35 to 7.45, and various systems are in place to keep that alkaline environment in balance. As you continue to read this article, you will discover the significance of ACV to human body’s pH+ level and how it can impact overall health when incorporated into your diet.
What is Apple Cider Vinegar?
Apple cider vinegar (ACV) is a type of vinegar made from crushed apples, yeast, and sugar. The fermentation process first starts with the fermentation of apple juice by yeast, which converts the sugars into alcohol. Then, bacteria are added to convert the alcohol into acetic acid, giving vinegar its characteristic taste and smell. Apple cider vinegar is commonly used as an ingredient in various dishes, such as salad dressings, pickles, and marinades.
The acidity of apple cider vinegar is attributed to its acetic acid content. Measuring around 4-5% acidity, apple cider vinegar is considered a safe level of acidity for culinary uses, such as canning and preserving foods. Additionally, ACV is believed to have antioxidant, antiviral, and antimicrobial properties; however, it is important to note that not all of these benefits are backed by scientific research.
With a pH level ranging from 2-3, apple cider vinegar is considered acidic. It is nutritionally similar to other types of vinegar, and it’s important to be aware of the potential risks and benefits associated with its consumption. When used in moderation, apple cider vinegar can be a flavorful and versatile addition to a variety of dishes.
Understanding pH
pH is a measure of how acidic or basic a substance is. It helps us to determine the properties and behavior of different solutions. In this section, we will discuss the concepts of acids and bases and the pH scale.
Acids and Bases
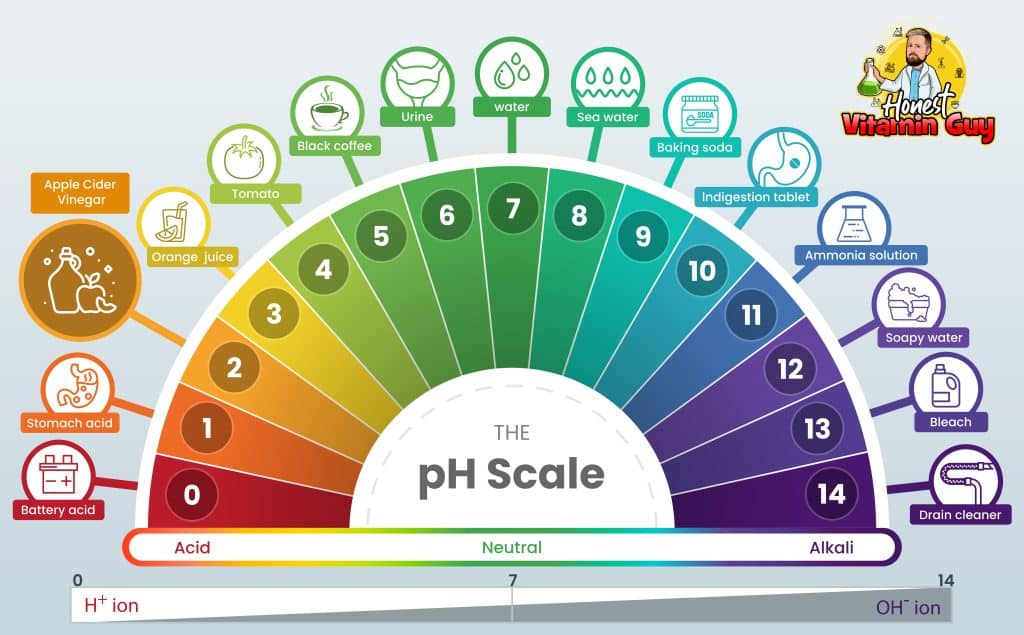
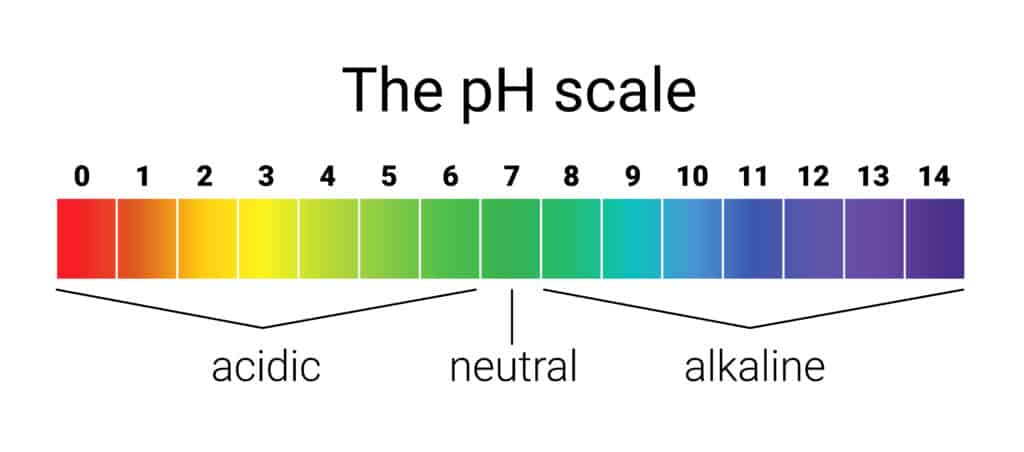
Acids are substances that have a pH less than 7, while bases have a pH greater than 7. Substances with a pH of 7 are considered neutral. Acids have the ability to donate protons (H+ ions) while bases can accept protons. In the context of apple cider vinegar, it is classified as an acidic substance because its pH falls within the range of 2-3.

pH Scale
The pH scale ranges from 0 to 14, with 0 being the most acidic, 14 being the most basic, and 7 being neutral. Here’s a simple breakdown of the pH scale:
0-6: Acidic
7: Neutral
8-14: Basic
It is important to note that the pH scale is logarithmic, meaning that a change in one pH unit represents a tenfold change in acidity or alkalinity. For example, a substance with a pH of 3 is ten times more acidic than a substance with a pH of 4.
Understanding the pH balance of substances like apple cider vinegar helps us to determine its properties and potential uses. With apple cider vinegar’s pH level ranging from 2-3, it is considered a mildly acidic substance. This information can be useful when assessing its potential health benefits and safety.

pH of Apple Cider Vinegar
Apple cider vinegar is considered to be an acidic substance. The pH of apple cider vinegar typically falls between 2 and 3, making it over 100 times more acidic than carbonated water with a pH of 5. Despite its acidity, some people believe that apple cider vinegar may have an alkalizing effect on the body when consumed, possibly because of its mineral content.
It is important to note that while the acidic nature of apple cider vinegar may contribute to some of its health benefits, it can also cause potential issues when not consumed properly. For example, consuming undiluted apple cider vinegar can lead to tooth enamel erosion and irritation of the throat due to its high acidity. To minimize these risks, it is recommended to dilute apple cider vinegar with water or other liquids before consumption.
In summary, the pH of apple cider vinegar is generally between 2 and 3, making it an acidic substance. Although acidic in nature, it may have potential health benefits due to its unique properties and contents. To fully reap the many possible health benefits, of apple cider, and avoid potential risks, it is essential to consume this vinegar in a safe and diluted manner.
Why does pH matter?
pH in the Human Body (Health Benefits)
The pH level in the human body is essential for maintaining good health. For example, the blood’s pH is tightly regulated at a slightly alkaline range of 7.35 to 7.45, to ensure proper cellular function and prevent various health issues. An imbalance in the the body’s pH levels can cause issues such as acidosis or alkalosis, which can be detrimental to one’s health. Acid imbalances may lead to fatigue, inflammation, and other symptoms.
Apple cider vinegar, with its acidic pH of 2-3, may help with indigestion and heartburn due to its potential ability to balance stomach acid levels. Additionally, some studies suggest that consuming apple cider vinegar may aid in weight loss and blood sugar control. However, it’s essential not to overconsume apple cider vinegar, as excessive amounts can have adverse effects.
pH in Cleaning

Knowing the pH of a cleaning solution, such as apple cider vinegar, is essential for determining its effectiveness in different cleaning tasks. Acidic substances like vinegar are effective at breaking down mineral deposits, cutting through grease, and disinfecting surfaces. For example, white distilled vinegar, a popular cleaning solution, has a pH of around 2.5, making it a useful natural cleaner.
Apple cider vinegar, with its pH of 2-3, offers similar cleaning benefits as other types of vinegar. However you buy apple cider vinegar, it is essential to dilute it with water before using it on surfaces, as concentrated vinegar can be both too much acid and harsh for some materials. By understanding and leveraging the pH of apple cider vinegar, you can make the most of its cleaning capabilities and help maintain a healthier living environment.
Comparing with Other Vinegars

When examining the pH levels of different types of apple cider vinegar supplements, it is important to compare apple cider vinegar to other commonly used vinegars. One of its closest counterparts is white vinegar, which has a similar acidity level.
Apple cider vinegar contains about five to six percent acetic acid, while white vinegar contains roughly five to ten percent. As a result, the acidity levels of both vinegars are quite comparable, with a pH value of 2–3.
Besides apple cider vinegar and white vinegar, there are other vinegars available with varying levels of acidity. For instance, balsamic vinegar and red wine vinegar offer different flavors and similar pH levels, making them suitable for various culinary applications.
It is crucial to consider the specific type of vinegar when determining its appropriate use, as each variety possesses unique properties and flavors that complement or enhance certain dishes.
Conclusion

After examining the various aspects of apple cider vinegar, it is clear that its pH level falls between 2-3, which classifies it as an acidic substance. This acidity can play a significant role in its various health benefits and applications, but also warrants caution due to potential side effects.
While some proponents claim that drinking apple cider vinegar can help to alkalize the body, there is limited scientific evidence to support these claims. Regardless, the acid content in apple cider vinegar has been linked to aiding digestion and other health benefits, but it remains crucial to strike a balance when incorporating it into one’s diet.
Incorporating apple cider vinegar should be done with moderation and consideration for potential side effects, such as tooth erosion, skin burns, and chemical burns. Like with any other supplement or dietary addition, consulting a healthcare professional for personalized advice is always the best practice.
Frequently Asked Questions
Is Apple Cider Vinegar Acidic?
Yes, Apple Cider Vinegar is acidic, with a pH of 2.5. This is on the acidic side of the pH scale’s neutral point at 7.
Is Apple Cider Vinegar Alkaline?
No, Apple Cider Vinegar is not alkaline. It has a pH of of 2.5, which is on the acidic side of the pH scale’s neutral point of 7. Above 7 is alkaline, but below is acidic.


